Thymosin Beta-4 and the Actin Pool: What TB-500 Actually Does at the Cellular Level
TB-500 is a fragment of thymosin beta-4, a 43-residue protein whose primary cellular role is sequestering G-actin monomers. The mechanism is unusual, the literature is dense, and the therapeutic implications are still being worked out.
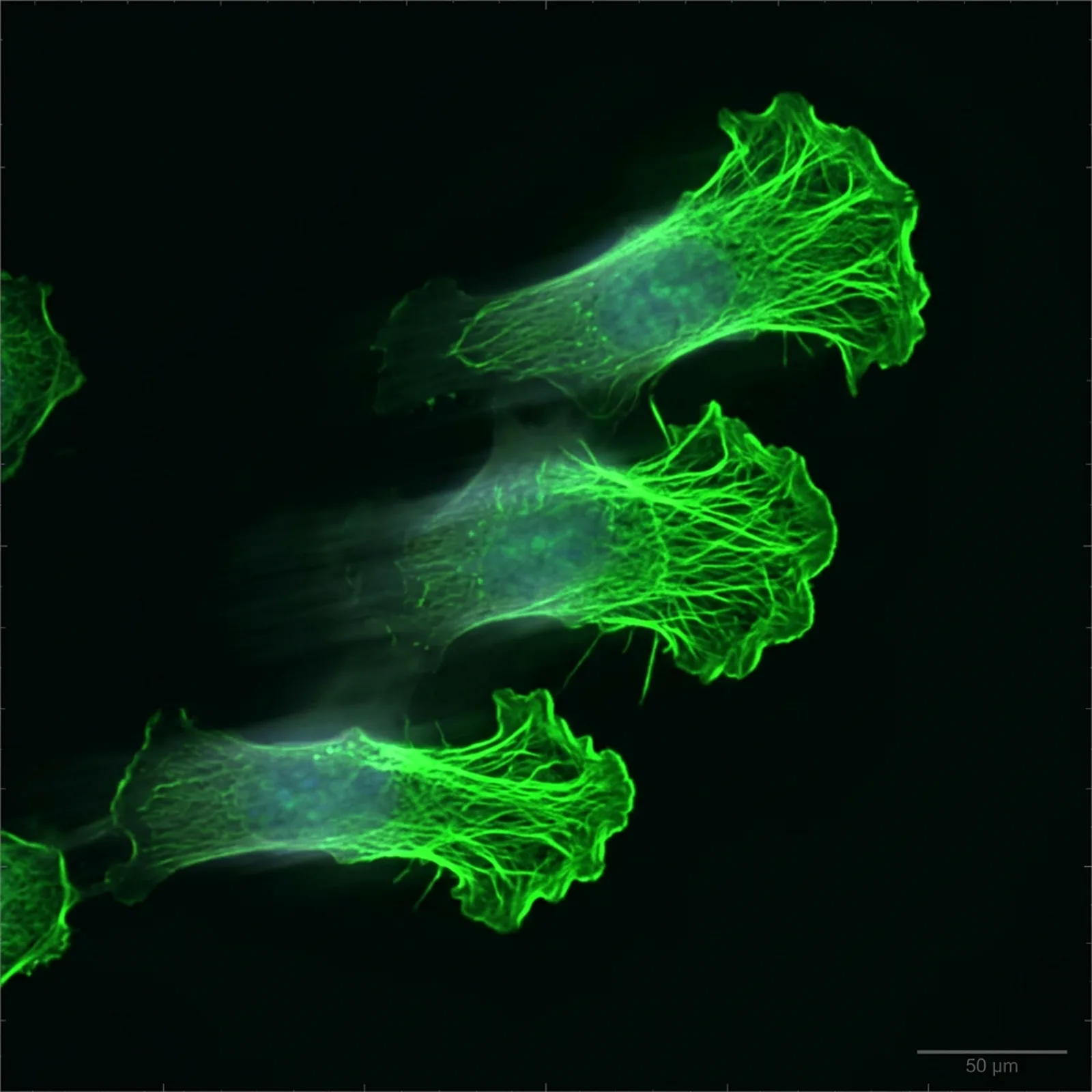
The Protein That Holds Half the Actin
If you tracked down every G-actin monomer in a vertebrate cell and asked where they all are, the answer would surprise you. About half of them are bound to a single small protein. That protein is thymosin beta-4. It's 43 amino acids long. It has no known enzymatic activity. Its sole job, as far as anyone can tell, is to hold G-actin monomers in solution and keep them ready for polymerization on demand.
This sounds like a passive role until you understand how dependent cells are on having that monomer pool available. Cell migration — the wound-edge crawl that closes lacerations, the metastatic shuffle of cancer cells, the macrophage chase after a pathogen — requires rapid, directional F-actin polymerization at the leading edge. The G-actin to fuel that polymerization has to come from somewhere. If the pool is exhausted, migration stops. If the pool is full, migration is fast.
Thymosin beta-4 is what keeps the pool full. Inside cells it binds G-actin in a 1:1 stoichiometry with high enough affinity to hold the monomers and low enough affinity that the actin can dissociate when the local F-actin pool needs to grow. It's a buffering protein, not a brake. And the wound-healing biology of TB-500 starts here.
The Malinda Wound Paper
The most-cited paper in the TB-500 literature is Malinda, Goldstein, and Kleinman in the Journal of Investigative Dermatology (1999). They showed two things that mattered. First, in a rat full-thickness wound model, topical thymosin beta-4 increased reepithelialization by 42% over saline controls at day 4 and 61% at day 7. Second, in cultured keratinocytes, Tβ4 accelerated migration with effects detectable at concentrations as low as 10 picograms per milliliter. The picogram sensitivity was striking — most peptide signaling effects in cell culture are in the nanogram-to-microgram range.
The mechanism they proposed tied directly back to actin biology. Tβ4 increased the available G-actin pool in keratinocytes, enabling faster F-actin polymerization at the migrating edge of the wound, accelerating coverage of the open dermis. Subsequent work added angiogenic effects (Tβ4 promotes endothelial cell migration through similar mechanisms) and anti-inflammatory effects (Tβ4 modulates macrophage polarization and reduces some inflammatory cytokine production). But the actin story is the foundation.
RegeneRx and the Drug Programs
The biotech company RegeneRx Biopharmaceuticals (now part of OncBioMune) ran clinical development of full-length Tβ4 for several indications through the 2000s and 2010s. The drug name varied by program — RGN-352 for the systemic injectable form used in cardiac trials, RGN-259 for the topical ophthalmic formulation used for dry eye, and others for wound applications.
The dry eye program, RGN-259, advanced furthest. It completed Phase II and Phase III trials with mixed results — some endpoints met, others not, with the FDA review process spanning multiple submissions. The cardiac program, in patients with acute myocardial infarction, produced positive Phase II signals on safety and on some efficacy measures, but the program didn't advance to Phase III. The wound-healing programs in pressure ulcers and venous stasis ulcers reported positive Phase II results that didn't lead to approval.
The pattern across these programs is consistent with a peptide that has real biological activity but operates with effect sizes that are difficult to power in modest trials. Twenty percent improvements in healing rates show up reliably, but the trial designs needed to convert those signals into FDA-grade efficacy demonstrations are large and expensive. RegeneRx ran out of capital before the larger Phase III programs could be fully resourced.
TB-500 vs Full-Length Tβ4
Researchers sometimes assume TB-500 is just a marketing name for thymosin beta-4. It isn't. TB-500 is a 17-amino-acid fragment that includes the actin-binding region of the full 43-residue parent protein. Most of the biological activities of full-length Tβ4 are reproduced by TB-500 in published head-to-head comparisons, but not all. Some immunomodulatory effects of full Tβ4 require regions of the protein outside the central fragment.
For wound-healing and cell-migration assays, TB-500 and full Tβ4 perform similarly. For experiments where the broader signaling pleiotropy of Tβ4 matters, full-length Tβ4 is the better tool. The synthesis cost difference between a 17-mer and a 43-mer is significant, which is why TB-500 dominates the research-peptide market while full Tβ4 is mostly used in academic labs running specific mechanistic comparisons.
The Stack Logic With BPC-157
The combination of BPC-157 and TB-500 is one of the most common research peptide stacks. The rationale is mechanistic, not synergistic in any rigorously demonstrated sense. BPC-157 promotes growth factor expression, vascular function, and tissue survival under stress. TB-500 maintains the actin pool that enables migration of repair cells into the injury site. The two operate at different stages of tissue repair: TB-500 helps cells get there, BPC-157 helps them stay alive and functional once they arrive.
There's no head-to-head trial of the combination versus either compound alone. The combination logic comes from mechanistic complementarity, not from a controlled comparison. Researchers running the stack should keep that distinction in mind — the case for using both is plausible, but the case for the combination being substantially better than either alone has not been rigorously tested.
Storage and Handling
TB-500 is a peptide of intermediate stability. Lyophilized at -20°C, it remains stable for years. Reconstituted in bacteriostatic water and stored at 2-8°C, it should be used within 30 days. Repeated freeze-thaw cycles degrade activity. Aliquoting on first reconstitution is standard practice.
For research applications, working concentrations in cell culture are typically 100 ng/mL to 10 µg/mL depending on the assay. For animal injury models, dose ranges in the published literature span 10 µg/kg to 1 mg/kg, with most protocols clustering around 100-500 µg/kg administered every few days. The Malinda 1999 paper and the various RegeneRx publications are the cleanest dose-response references in the human and animal literature respectively.
Sources: Malinda, Goldstein, Kleinman, J Invest Dermatol, 1999; Goldstein and Hannappel, Annals NY Acad Sci, 2007; Smart et al., Nature, 2007 (cardiac Tβ4); Sosne et al., Cornea, 2010 (dry eye); RegeneRx clinicaltrials.gov filings, 2008-2018; Goldstein, Hannappel, Sosne, Kleinman review, Expert Opin Biol Ther, 2012.
Frequently Asked Questions
Is TB-500 actually thymosin beta-4?
<a href="/product/tb-500-5mg">TB-500</a> is a fragment of thymosin beta-4. The full thymosin beta-4 protein is 43 amino acids; TB-500 is the central 17-residue active fragment. The two are not identical, but TB-500 retains the actin-binding region of the parent protein and reproduces most of the biological activities seen with full-length Tβ4 in published comparison studies.
What does 'G-actin sequestration' mean and why does it matter?
Actin exists in two forms in the cell: monomeric G-actin (free in the cytoplasm) and polymeric F-actin (organized into filaments). For a cell to migrate, it has to rapidly polymerize F-actin at the leading edge while depolymerizing it at the rear. This requires a substantial pool of G-actin available on demand. <a href="/product/tb-500-5mg">Thymosin beta-4</a> is the major G-actin sequestering protein in vertebrate cells — it binds free G-actin and holds it in a polymerization-ready state. Without an adequate G-actin pool, cell migration stalls.
What did Malinda 1999 actually show?
Malinda, Goldstein, and Kleinman published in the Journal of Investigative Dermatology (1999) a study showing that <a href="/product/tb-500-5mg">thymosin beta-4</a> increased wound reepithelialization by 42% over saline controls at day 4 and up to 61% at day 7 in a rat full-thickness wound model. They also demonstrated keratinocyte migration acceleration in cell culture, with effects detectable at picogram concentrations. The paper has been cited heavily and remains foundational for the wound-healing case.
Has TB-500 / Tβ4 been in human clinical trials?
Yes. RegeneRx Biopharmaceuticals ran Phase I and Phase II trials of full-length <a href="/product/tb-500-5mg">Tβ4</a> (under the codes RGN-352 and RGN-259) for indications including pressure ulcers, venous stasis ulcers, dry eye disease, and acute myocardial infarction. The dry eye program advanced to Phase III. The cardiac program produced positive Phase II safety and efficacy signals but didn't progress further. Trial data are publicly available through clinicaltrials.gov and RegeneRx's regulatory filings.
What's the half-life difference between TB-500 and full Tβ4?
<a href="/product/tb-500-5mg">TB-500</a>'s circulating half-life in animal studies is approximately 3 hours, similar to full-length Tβ4 within experimental error. Both molecules are subject to relatively rapid renal clearance. The shorter overall length of TB-500 doesn't dramatically change half-life because the limiting clearance mechanism is renal filtration, which scales weakly with the size differences between a 17-mer and a 43-mer at this molecular weight range.